Historical Context: How It All Began
At the beginning of the 20th century, scientists were studying how hot objects, such as stars, emit energy.
A key scientist was Max Planck.
He discovered that energy is not emitted continuously, but in small packets called quanta.
To understand it more easily:
Before, people thought energy was like water flowing continuously.
Planck said it is actually more like coins that are given one by one.
This discovery marked the beginning of quantum mechanics.
The Problem of Measuring Particles
Then, in 1927, Werner Heisenberg introduced an idea that would change physics.
To know exactly where a particle is, we have to observe it.
But observing extremely small particles (like electrons) changes their state.
For example:
You want to see a mosquito in the dark → you use a flashlight → the mosquito moves.
The same happens with an electron:
when you try to see it, you push it (because of the photons of light) and change its velocity.
The Formula
Heisenberg wrote it like this:
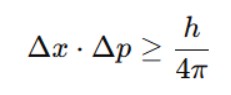
Δx = uncertainty in position
Δp = uncertainty in velocity or momentum
h = Planck’s constant, a very tiny number
Planck’s constant is the number that indicates how much energy each packet has, depending on the frequency of the light.
This means:
The product of the uncertainties can never be smaller than a certain limit.
If you make Δx very small (a very precise measurement of position), then Δp must be large (large uncertainty in velocity).
And vice versa.
The Uncertainty Principle
To measure the position of a particle, you must interact with it, for example by hitting it with light (photons).
If you use very precise light to know exactly where it is → the photons give it a strong “push” → its velocity changes a lot.
If you use less intense light → you don’t change its velocity as much → but you cannot know exactly where it is.
Therefore:
It is impossible to know the exact position and the exact velocity of a particle at the same time.
Measuring one of them too precisely interferes with the other, because observing very small particles always disturbs them.
This is not a problem with instruments, it is a real property of nature.
How Quantum Mechanics Explains It
Quantum physics introduced the idea that particles do not have exact values of position and velocity.
Instead, they are described by a quantum state.
This state tells us the probabilities of finding the particle in certain positions and with certain velocities.
It is as if the particle exists in several places at the same time until it is measured.
Example:
We do not say:
“The electron is exactly here.”
Instead we say:
“There is an 80% probability it is here and a 20% probability it is there.”
Summary
- Before, scientists believed energy and matter could be measured exactly.
- Planck discovered quanta, small units of energy.
- Heisenberg discovered that position and velocity cannot both be measured with perfect precision at the same time.
- Particles are described using probabilities, not exact values.
- This is not human measurement error, it is simply how nature behaves at the quantum level.
- In the world of very small particles, nature itself is uncertain:
the more precisely you know the position of a particle, the less precisely you know its velocity, and vice versa.
When we measure things in science, we usually obtain a range of possible values, like when measuring temperature.
Heisenberg discovered that in the world of particles this is not just a measurement limitation:
nature itself prevents us from knowing the exact position and velocity at the same time.
Leave a comment